Improving epilepsy care: HBP researchers involved in major clinical trial
24 October 2018
In a world’s first, personalized brain modelling is providing the basis for a large-scale clinical trial in epilepsy. The approach could bring the first breakthrough in decades for drug resistant epilepsy patients undergoing brain surgery.
Press Release 19 October 2018
In a world’s first, personalized brain modelling is providing the basis for a large-scale clinical trial in epilepsy. The approach could bring the first breakthrough in decades for drug resistant epilepsy patients undergoing brain surgery.
The project EPINOV, short for “Improving EPilepsy surgery management and progNOsis using Virtual brain technology”, is testing the use of brain modeling to improve epilepsy care in a large clinical study. The project is based on the brain simulation technology “The Virtual Brain”, that HBP scientist Viktor Jirsa has developed with collaborators. To help clinicians plan surgery strategies, the scientists create personalized brain models of patients and simulate the spread of abnormal activity during epileptic seizures. Two pilot studies showed promising results for the approach. Now, this personalized brain modelling approach is providing the basis for a large-scale clinical trial in 11 French hospitals. A consortium has taken up preparatory work at the beginning of this year, with the clinical testing planned to begin in January 2019.
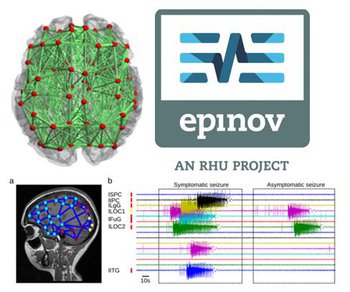
Epilepsy affects about one percent of the population and comes with debilitating seizures, caused by waves of synchronized brain activity propagating through the brain. While for many patients these seizures can be controlled by drugs, almost a third of the patients are drug resistant. For them, only surgery remains as an option in which the epileptogenic zone – the area or network from which the seizure activity emerges and then propagates throughout the brain – is removed.
“The challenge in planning these surgeries is to localize the epileptogenic zone as precisely as possible”, Viktor Jirsa explains. The scientist is Director of Inserm’s Institut de Neurosciences des Systèmes (INS) at Aix-Marseille University and Director of Research at the CNRS. In HBP he is the Deputy Leader of the research area of Theoretical Neuroscience and Scientific Leader of a project developing a multimodal and multi-scale human brain atlas.
During pre-surgery assessment, clinicians mostly rely on measurements from electrodes in the brain or from EEG, but interpreting these data sources can be difficult. As a result, surgery outcomes are often unpredictable, with seizures persisting in almost half of the patients post-surgery. “This success rate has largely stayed the same the last 30 years, there really has been somewhat of a stalemate in this. We hope our approach can finally improve the odds for patients”, Jirsa says.
At the core of the new approach is a simulation engine called The Virtual Brain. First, a personalized brain model is created from data of the individually measured anatomy, structural connectivity and brain dynamics for each patient. Through a series of steps (see below “How it works”) this is turned into a dynamic network model, on which the seizure propagation can be simulated. High Performance Computing enables the personalization of the brain network models through machine learning. This “brain avatar” is customized to the individual patient and allows testing and estimates about different strategies during surgery preparation. The essential concept underlying this approach is the exploitation of the large-scale network properties of a patient’s brain. A detailed account of this work can be read here: Epilepsy: Building Personalised Models of the Brain (HBP news item, Nov 2017)
How it works:
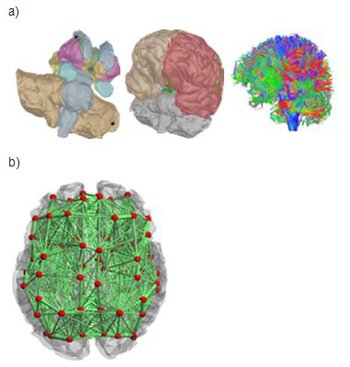
Virtualisation of a personalised brain model. a) Non-invasive imaging (MRI, CT) provides personalised images of a human subject's brain. From these images, Jirsa's team reconstructs subcortical and cortical surfaces (top left and middle) and connectivity (top right). These elements are aligned and coregistered in the same coordinate system. A parcellation of the surfaces breaks the brain down into network nodes, which are then connected by the personalised connectome. b) Network nodes and connectivity are organised in 3D physical space, providing a three-dimensional avatar. Once each network node is equipped with a mathematical representation of a neuronal population model, the brain model is functional and computer simulations generate brain model activity and its associated neuroimaging signals.
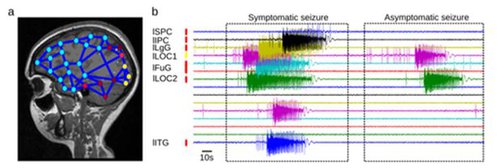
Virtual Epileptic Patient. a) The Epileptogenic Zone (EZ) is traced out in a patient's brain model, where a heat map shows the spatial distribution of epileptogenicity (from low (cold colors) to high (hot colors) epileptogenicity). The heat map is either based on the clinician's hypothesis or derived from the application of machine learning methods to the model and empirical SEEG data. b) The patient's brain model generates spontaneous seizure activity mimicking the various seizure propagation patterns and its dynamic repertoire of symptomatic and asymptomatic seizures.
“In a small cohort of retrospective surgery patients we were able to demonstrate that the predictions of the patient’s brain model correlate better with positive surgery outcome than the clinicians’ predictions”, the scientist explains, “and other labs have confirmed our results independently.” Now Jirsa, his team and collaborators are preparing the biggest step yet: To run a full-scale multi-centric clinical trial of almost 400 prospective patients, involving clinical and neuroimaging experts, theorists like Jirsa, and also industry researchers from the projects partner Dassault Systémes, which develops a first prototype of the modeling and simulation software. If the approach proves useful, this software could later be provided to hospitals and Epilepsy centers around the world.
“There is a very big responsibility”, Jirsa says “and at the same time it’s exciting that we have this chance to improve clinical practice and ultimately patients’ lives.” In half of the cases, the surgeons will have information from the Epilepsy-model in their staff meetings, where therapeutic interventions are planned. The study follows a blind random design, where half of the patients will be operated taking the model predictions into account and the other half will not. “After four years, the statistics will show us hopefully to what degree the model predictions changed the surgery practice and outcome”, Jirsa says.
Jirsa and his close collaborator Randy McIntosh at Baycrest Center Toronto started building The Virtual Brain as an open source brain network simulator from 2010 on, using neuronal population models and structural information from neuroimaging. Over the last 5 years and in large part within the framework of HBP, this connectome-based large-scale brain modeling approach was adapted to applications in Epilepsy. Many elements of the modeling in this clinical trial have been developed within the Theoretical Neuroscience research area of HBP over the last five years, and published in many high-profile publications by the team.
For the coming years there are extensive plans to expand this work within HBP. Earlier this year, The Virtual Brain joined HBP as a Co-Design Project under the leadership of Petra Ritter at Charité University Hospital Berlin with the goal of connecting The Virtual Brain to HBP´s Brain Simulation Platform. (See July 3 press release: The Virtual Brain neuroinformatics platform joins the EU's Flagship Human Brain Project)
“The simulation aspect in HBP gives us the opportunity to bring together our macroscale-approach to modelling and simulation with a range of different simulation-based approaches. Scientifically, that lets us better understand how phenomena on the micro-level might translate to higher order phenomena on the meso- and macro-scale, and can open possibilities of a “consistency test” between the models.”
The scientists are also continuing their activities on clinical modeling for dementia, stroke and epilepsy in collaboration with experts that the HBP brings together. If the approach that is tested in the EPINOV-trial succeeds, it would be the first modelling-based case of personalized medicine that makes the jump from research to clinical practice – the outcome thus will be a signal for the whole field.
For Jirsa seeing his work as a theoretical scientist have this potential impact has been the result of a unique convergence: “When we began, concepts like Connectomics where not yet around, it was a very new field. That we have come to this point was made possible by clinical expertise and our activities on The Virtual Brain and the Human Brain Project all coming together – right place, right time, right people."
Contact:
Prof. Viktor Jirsa
Email: viktor.jirsa@univ-amu.fr
tel : ++33 (0)4 91 32 42 51
Publications:
Proix T, Jirsa VK, Bartolomei F, Guye M, Truccolo W.: Predicting the spatiotemporal diversity of seizure propagation and termination in human focal epilepsy. Nat Commun. 2018 Mar 14;9(1):1088. doi: 10.1038/s41467-018-02973-y.
Proix T, Bartolomei F, Guye M, Jirsa VK (2017) Individual brain structure and modeling predict seizure propagation. Brain 140 (3): 641-654 https://www.ncbi.nlm.nih.gov/pubmed/28364550
Jirsa VK, Proix T, Perdikis D, Woodman MM, Wang H, Gonzalez-Martinez J, Bernard C, Bénar C, Chauvel P, Bartolomei F (2016) The Virtual Epileptic Patient: Individualized whole-brain models of epilepsy spread. Neuroimage doi :10.1016/j.neuroimage.2016.04.049



