Computational Modelling in Hippocampal and Cortical Neurons
26 June 2020
A new approach can correlate changes in the time course of individual synaptic events with the changes in specific biomolecular pathways after a single-protein block: the case of Gephyrin.
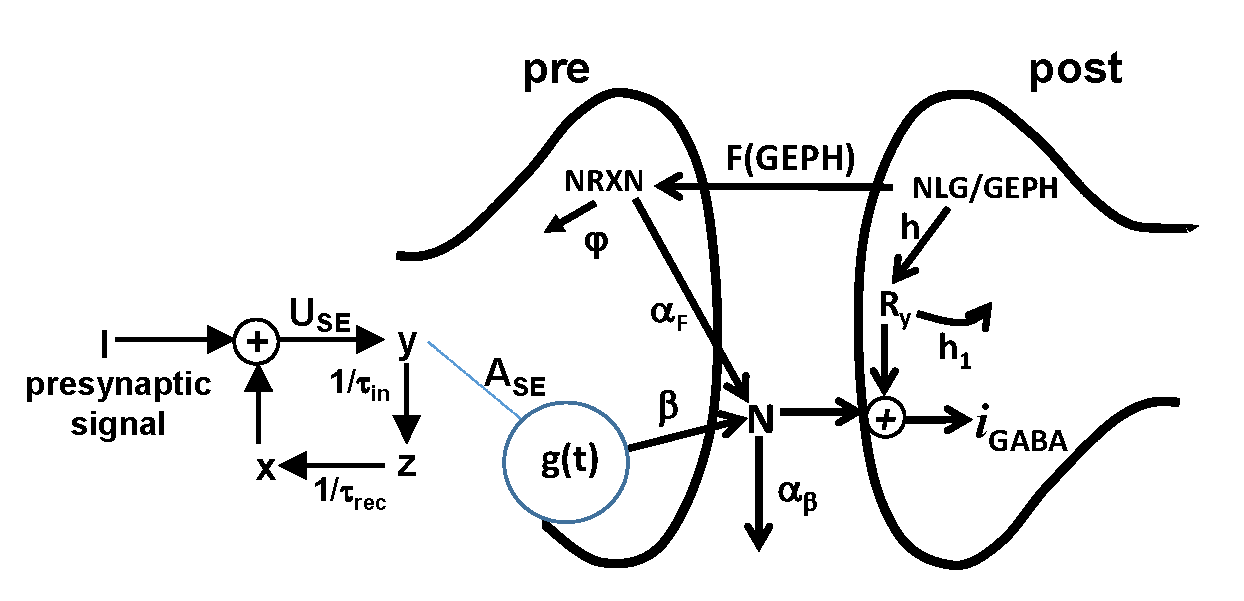
GABAergic transmission regulates several processes involved in cognitive functions. By anchoring synaptic receptors just opposite to release sites, the scaffold protein gephyrin plays a key role in these processes. In addition, by regulating GABAA receptor trafficking, gephyrin contributes to maintain, at the network level, an appropriate balance between Excitation (E) and Inhibition (I), crucial for information processing. An E/I imbalance leads to neuropsychiatric disorders such as epilepsy, schizophrenia and autism.
An international team of researchers working in the Human Brain Project, by analysing experimental recordings of synaptic events with a computational model, has been able to correlate the amplitude and time course of individual events with changes that may occur in the main subcellular pathways involving gephyrin-dependent synaptic transmission. The findings, published in the Frontiers in Cellular Neuroscience Journal, increase our knowledge on the basic mechanisms underlying inhibitory trans-synaptic signalling. In particular, it has been revealed that interfering with gephyrin function affects not only the organisation of postsynaptic receptor clustering but also presynaptic factors including neurotransmitter release, reinforcing the intriguing idea that gephyrin can trans-synaptically organise inhibitory synapses, thus representing an important synaptic "molecular hub”.
The scientists from the two laboratories involved in the project used a new approach, involving a computational method to fit and analyse spontaneous synaptic events recorded under different gephyrin interference experimental protocols. More broadly, this approach can be useful to analyse experiments designed to block any given single protein of the inhibitory post synaptic density.
The results provided in the paper include contributions from the research groups of Antonino Cattaneo, Enrico Cherubini and Silvia Marinelli, from the European Brain Research Institute (EBRI, Roma, Italy), and Michele Migliore, from the Institute of Biophysics of the National Research Council (CNR, Palermo, Italy). The project has received funding from the Human Brain Project.
Further reading:



