New technique enables imaging of intact mouse brains at subcellular resolution
23 August 2021
Understanding the complex structure and functioning of the brain is one of the greatest scientific challenges of the 21st century. Part of the challenge is the analysis of brain architecture at cellular and subcellular levels. A team of researchers from Italy, the UK and Germany have developed a new microscopy technique called Rapid Autofocus via Pupil-split Image phase Detection (RAPID) that enables imaging of voluminous samples at higher resolutions than previously possible – and has applications beyond neuroscience. Their work has recently been published in Nature Methods and was carried out within the European Flagship Human Brain Project (HBP). The lead developers of RAPID from the LENS laboratory in Florence are long-time contributors to the HBP’s brain mapping and atlasing work.
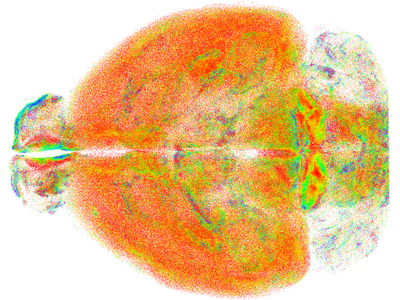
Point cloud of all somatostatin-positive neurons in the mouse brain © LENS
The authors have successfully used RAPID to obtain images of intact mouse brains at resolutions that allowed them to study subcellular structures. They were able to observe minute neural processes that have previously not been visible on a brain-wide scale. The researchers also applied RAPID to study how special types of neurons are spatially arranged in the brain and to analyse the three-dimensional shape of microglia in different brain regions. “The new technique will have a significant impact in the field of neuroscience by enabling researchers to quantitatively analyse the architecture of the brain at a subcellular level,” said Ludovico Silvestri.
Up until now, analysis of whole mouse brains under the microscope had only been possible at low resolution. "The currently employed method of light-sheet microscopy combined with chemical protocols capable of rendering biological tissues transparent, fails to maintain high resolution in samples larger than a few hundred microns," said first author of the study, Ludovico Silvestri from the University of Florence. In light-sheet microscopy, only a thin layer of the sample, the focal plane, is illuminated with light. However, if the sample is too thick it acts like an optical lens which disrupts the alignment of the microscope, shifting the focal plane of the microscope’s objective away from the light sheet. This shift leads to blurry images.
The team has developed RAPID to overcome this challenge. The method is inspired by the autofocus system found in reflex cameras. When the object is defocused, rays of light passing through specific parts of the objective intersect the image plane at different lateral positions. In RAPID, incoming rays of light are separated with a wedge plate and an auxiliary camera is used to take a second, laterally displaced image with a slight variation in focus. The lateral displacement between the two images provides information about the focal state of the microscope (it is directly proportional to it) and can then be used to stabilize the focus. The team has optimized the microscope’s set-up to achieve autofocussing of images within a few hundred milliseconds while taking images of their sample in parallel.
The authors stress that RAPID does not require advanced expertise in optics. To set up the system, only off-the-shelf optical components and well-established algorithms are needed.
They also demonstrate that RAPID can be combined with multiple methods for tissue clearing and staining and is not limited to light-sheet microscopy. “While we originally invented RAPID for light-sheet microscopy, this autofocusing technology is actually suitable for all wide-field microscopy techniques. It is very versatile and sample agnostic with multiple applications beyond neuroscience,” said co-author Caroline Müllenbroich from the University of Glasgow.



