EBRAINS Workshop
HBP Partnering Projects Meeting: status quo and outlook
5 – 7 September 2022 | Nijmegen, Netherlands
Download Final Programme 3.0 MB
Check out the recordings of the lectures of this workshop
About
 © Liam Read
© Liam Read
By plane
Nijmegen doesn't have its own airport. If you need to go by plane, the best route would be to go via Amsterdam Schiphol. This airport is one of the busiest airports in Europe and is easy to reach from many European and American cities.
Amsterdam is only 89 km from Nijmegen. Going from Amsterdam to Nijmegen by train takes less than 90 minutes. Trains run multiples every hour.
By train
The Netherlands is well connected by train. The principal Dutch railway operator is Nederlandse Spoorwegen. They operate all of Netherlands and on their website train journeys starting abroad can also be booked. NS also offers a number of travel tickets for the Netherlands. All information can be found on their website.
The main train station in Nijmegen is called Nijmegen Railway Station (Centraal Station Nijmegen). It is very well connected with other Dutch cities via the Intercity network. Trains between Amsterdam and Nijmegen run twice an hour.
Other train stations in Nijmegen are Nijmegen Dukenburg, Nijmegen Heyendaal, Nijmegen Lent and Nijmegen Goffert.
By car
Nijmegen is located at the highway A73 and therefore easily reachable by car.
The speed limits on Dutch streets are as follows:
- 50 km/h inside built-up areas
- 80 km/h outside built-up areas
- 100 km/h on expressways
- 100 km/h from 6:00 - 19:00 and 130 km/h from 19:00 - 06:00 on motorways
The venue is located in Berg en Dal, which is a village located to the southeast of Nijmegen.
By car
The car ride to Berg en Dal takes 11 minutes. From Nijmegen, just take the Berg en Dalsweg to Berg en Dal.
By public transport
Take the Breng Bus 8 in the direction Berg en Dal and get off at the stop De Meerberg, Berg en Dal. The bus ride will take approximately 23 minutes. The venue is located a 12 minutes walk away from the bus stop.
The hotels, which are closest to the venue, are Fletcher Parkhotel Van Monte and Fletcher Hotel-Restaurant Erica. They are also part of the same hotel chain as the venue. You can book a room via the hotel websites.
As of April 2022, travellers from the EU, the Schengen area, as well as countries that participate in the EU travel rules scheme, are allowed to freely enter the Netherlands. Travelers neither have to get tested nor be vaccinated.
In the Netherlands, all mask mandates have been lifted. Masks only have to be worn in airports and on airplanes.
The Netherlands has a moderate maritime climate. The main features of this are mild winters and cool summers. Rain is very common.
The average temperature in autumn is 10°C. It is also the season with the most rain.
Language
The official language of the Netherlands is Dutch. Dutch is a West Germanic language and thus closely related to German and English.
Most Dutch people speak very good English. In addition, many also speak German.
Currency
The currency in the Netherlands is Euro.
Time
The Netherlands is part of the CET (Central European Time). From April to October, the Netherlands is part of the CEST (Central European Summer Time). These time zones are UTC+1 and UTC+2 respectively.
Electricity
In the Netherlands the power plugs and sockets are of type C and F. These are the power plugs common in Europe. The standard voltage is 230 V and the standard frequency is 50 Hz.
Please note that the information provided on this site has been obtained from several different sources and therefore the organisers cannot accept any responsibility for errors therein.
As the Human Brain Project is in its final phase, it was time for Partnering Projects to convey the scientific community the tour-de-force they have made in furthering knowledge concerning various aspects of Brain Research. This EBRAINS Workshop offered Partnering Projects an international pedestal to showcase their major achievements to the Human Brain Project Partnering environment and the wider scientific community. The Workshop was open for scientists from various fields of Neuroscience research, and was hosted by the annual Donders Cognitive, Brain & Technology Summer school (DCBT 2022) promoting a shared systems-level understanding of the functional architecture of the brain and its possible emulation in artificial systems. The event was designed as a platform for Partnering Projects to exchange about their scientific findings, extend their collaborative network and engage into open discussions with EBRAINS representatives about the transition from the Human Brain Project to EBRAINS. It was a three-day event hosted at the Fletcher Landgoed Hotel Holthurnsche Hof (Nijmegen, the Netherlands). This created new communities of interested scientists that can critically contribute, as user or developer, to the evolution of EBRAINS as a sustainable infrastructure for the wider scientific community.
Registration
Registration is closed.
Call for submissions
We invited original, high-quality submissions describing innovative research addressing the topics of brain structure, function and cognition in health and disease, networks, brain simulation and modelling, neuromorphic computing, neurorobotic applications and neuroethics. The contributions could emphasise theoretical or empirical aspects. We particularly encouraged submissions exploiting computational models for a mechanistic understanding of brain function and cognition.
Poster session information
More information was sent to applicants upon acceptance of their poster abstract submission. The best poster presenter will receive an award (Best Poster Award certificate and a 500 euros prize in the form of travel reimbursement).
Call for submissions is closed.
If you have any questions, please contact workshop.edu@humanbrainproject.eu
Authors were informed about acceptance/rejection of their poster until 26 July 2022.
Picture gallery
Final Programme
| 08:30 - 09:00 | Registration & Coffee |
| 09:00 - 09:30 |
Welcome & Introduction Julien Vezoli, Paul Verschure, Marie-Elisabeth Colin |
| 09:30 - 10:30 |
Plenary Session I: Primate Brain Specifics Layer-specific characterization and modeling of fronto-parieto-visual dynamics in primate cortical networks | Bjørg Kilavik (Aix-Marseille University) & Sacha van Albada (Forschungszentrum Jülich) Comparative Investigation of the Cortical Circuits in Mouse, Non-human primate and Human | Julien Vezoli (ESI) |
| 10:30 - 11:00 | Coffee Break & Poster Session |
| 11:00 - 12:00 |
Plenary Lecture Deep brain stimulation of the thalamus restores signatures of consciousness in a non-human primate model | Béchir Jarraya (CEA Paris-Saclay, Centre NeuroSpin) |
| 12:00 - 13:00 | Lunch Break |
| 13:00 - 14:30 |
Plenary Session I: Primate Brain Specifics Computational analysis of primate vision | Simo Vanni (Helsinki University Hospital) | Slides Integrated effects of multiple Attentional Control signals in the primate brain | Elisa Santandrea (Verona University) | Slides Development of cortical multisensory integration mechanisms at micro- and macro- scales during normal and pathophysiological conditions | Conrado A. Bosman (University of Amsterdam) | Slides |
| 14:30 - 15:00 |
Implementation of the EBRAINS NHP Atlas and open discussion Wim Vanduffel (KU Leuven) | Slides |
| 15:00 - 15:30 | Coffee Break |
| 15:30 - 17:00 |
Parallel sessions: EBRAINS Atlases service | Timo Dickscheid (Forschungszentrum Jülich) EBRAINS Data & knowledge service | Ingrid Reiten (University of Oslo) |
| 17:00 - 17:30 | Summaries by rapporteurs & concluding remarks |
| 09:00 - 09:30 |
The EBRAINS Research Infrastructure from an HBP perspective: Status and Outlook EBRAINS RI, the future (and how to cooperate) |
| 09:30 - 10:30 |
Plenary Lecture Chemistry of the adaptive mind: lessons from dopamine | Roshan Cools (Donders Institute for Brain, Cognition and Behaviour) |
| 10:30 - 11:00 | Coffee Break & Poster Session |
| 11:00 - 12:00 |
Plenary Session II: Brain Circuits & Simulation The Pattern of Cellular Activity in the CA1 of the Hippocampus after Spatial Learning Depends on the Time Interval between Training Sessions | Eleonora Centofante (Sapienza Università di Roma) | Slides Map of dopamine 1- and 2-receptor positive cells in the developing mouse forebrain: perceived fit of registered atlas across sex, age, and regions | Jee Hyun Kim (Deakin University) |
| 12:00 - 13:00 | Lunch Break |
| 13:00 - 14:30 |
Plenary Session II: Brain Circuits & Simulation Project FIIND: Ferret Interactive Integrated Neuro Development Atlas | Roberto Toro (Institut Pasteur) SENSEI: uncovering neuron structure using hard and soft approaches | Nicola Vanello (University of Pisa) | Slides Integrative Frameworks in Single-cell Connectomics | Nestor Timonidis (Donders Institute for Brain, Cognition and Behaviour) | Slides |
| 14:30 - 15:00 |
From Human Brain Project to EBRAINS RI: coordinating the technical aspects of the transition Amaryllis Raouzaiou (Athena Research and Innovation Center) | Slides |
| 15:00 - 15:30 | Coffee Break |
| 15:30 - 17:00 |
Parallel sessions: EBRAINS Brain modelling & simulation service | Marmaduke Woodman (Aix-Marseille University) Massive Computing applied to EBRAINS | Lena Oden (Forschungszentrum Jülich) |
| 17:00 - 17:30 | Summaries by rapporteurs & concluding remarks |
| 09:00 - 10:00 |
Plenary Lecture Exploiting individual differences to inform computational models of dopamine in reinforcement learning | Mehdi Khamassi (Institute of Intelligent Systems and Robotics) |
| 10:00 - 10:30 | Coffee Break |
| 10:30 - 12:00 |
Plenary Session III: Applied Brain Models MULTI-LATERAL: Multi-level Integrative Analysis of Brain Lateralization for Language | Clyde Francks (Max Planck & Donders Institute & Radbound University) | Slides SMART BRAIN: Advanced Morphological Reconstruction of Human Brain Tissue by Multimodal Fusion of Multiscale Optical Imaging Technologies | Jonathan Mapelli (University of Modena and Reggio Emilia) | Slides CerebNEST: A Bioinspired Multiscale Modelling of The Cerebellar Network | Alessandra M. Trapani (Politecno di Milano) | Slides |
| 12:00 - 13:00 | Lunch Break |
| 13:00 - 15:00 |
Plenary Session III: Applied Brain Models Responsible Research and Innovation in Applied Brain Models | Mayen Cunden (De Montfort University) | Slides Efforts on the integration and large-scale simulation of a whole-brain model led to the development of a collaborative framework for data-driven modeling | Carlos Enrique Gutierrez (Okinawa Institute of Science and Technology) | Slides The Virtual Brain Cloud (TVB-Cloud) | Petra Ritter (Charité University Medicine Berlin) The RGS@HOME project: Scaling ICT based neurorehabilitation to personalized 24/7 home care | Paul Verschure (Donders Institute for Brain, Cognition and Behaviour) |
| 15:00 - 15:30 | Coffee Break |
| 15:30 - 16:30 |
Closing Plenary Lecture Chair: Julien Vezoli (Ernst Strügmann Institute (ESI) for Neuroscience in Cooperation with Max Planck Society & Paul Verschure (Donders Institute for Brain, Cognition and Behaviour) Enabling Complex Simulations with Multilevel Health Data | Petra Ritter (Charité University Medicine Berlin) |
| 16:30 - 17:00 |
Open Discussion: Workshop wrap-up & next steps Steven Vermeulen (EBRAINS), Paul Verschure (Donders Institute for Brain, Cognition and Behaviour), Julien Vezoli (Ernst Strügmann Institute (ESI) for Neuroscience in Cooperation with Max Planck Society) |
Confirmed Speakers
|
Ingvild Bjerke | University of Oslo |
Lena Oden | Forschungszentrum Jülich
|
Abstracts
Development of cortical multisensory integration mechanisms at micro- and macro- scales during normal and pathophysiological conditions
In this talk we will present how the DOMINO projects aim to investigate, with a multi-species and multi-scale approach, the hypothesis that impairments in multisensory integration are a key characteristics of autism-spectrum disorders. The results of the experiments that we are performing in animal models and human participants will contribute to the development of a computational model of multisensory integration and crossmodal plasticity that we aim to integrate within the EBRAINS platform.
THE PATTERN OF CELLULAR ACTIVITY IN THE CA1 OF THE HIPPOCAMPUS AFTER SPATIAL LEARNING DEPENDS ON THE TIME INTERVAL BETWEEN TRAINING SESSIONS
Eleonora Centofante1,2, Luca Fralleoni1, Carmen A. Lupasco2, Michele Migliore2, Arianna Rinaldi1, Andrea Mele1
1Department of Biology and Biotechnology ‘C. Darwin’-Centre for Research in Neurobiology ‘D.Bovet’, Sapienza University of Rome, Italy; 2Biophysics Institute, National Research Council, IBF-CNR, Palermo, Italy.
Training with long inter-session intervals, termed spaced or distributed training, has long been known to be superior to training with short intervals, termed massed training.In the present study we compared c-fos expression after massed and distributed training protocols in the spatial version of the MWM to outline possible differences in the learning induced pattern of neural activation in the dCA1 of hippocampus in the two training conditions. Two are the main findings yield:1. Distinct sub-regional distribution of c-fos positive cells after massed and distributed training; 2. Increased clustering of cell activity in mice trained with the distributed versus the massed protocol. Moreover, using a bagged tree algorithm we found that differences in the number of c-fos positive cells and their location in the dCA1 could be predictive of the training protocol used.
Interestingly our finding demonstrated that increased time lags between learning opportunities increased neuronal activity in the septo-caudal domain of the CA1. Parallel to this we found that although the overall number of c-fos expressing cells did not differ in the massed compared to the distributed group, mice trained with the distributed protocol show increased clustering of labelled cells in the brain region analyzed. These results suggest that the topographic organization and the spatial location of learning activated cell assemblies might be critical to promote the increased stability of the memory trace induced by distributed training, similarly to what has been found after prolonged training.
Roshan Cools is Principal Investigator at the Donders Institute for Brain, Cognition and Behaviour and Professor of Cognitive Neuropsychiatry at the Radboud University Medical Center. She is an expert in the chemical neuromodulation of human cognition and motivation, focusing on the functions of dopamine and serotonin. She completed her undergraduate degree in Experimental Psychology at the University of Groningen, Netherlands, in 1998. She then moved to Trevor Robbins’ lab at the University of Cambridge, UK, for an MPhil degree (1999), a PhD degree (2002), in cognitive neuroscience, a St John’s College Junior Research Fellowship (2002-2006) and a Royal Society Dorothy Hodgkin Research Fellowship (2002 – 2006). She spent two post-doc years at UC Berkeley working with Mark D’Esposito from 2003, before moving back to Cambridge in 2005, where she obtained a Royal Society University Research Fellowship (2006 -2007). In November 2007 she returned to The Netherlands, where she now runs her Motivational and Cognitive Control group. She is a member of the Royal Netherlands Academy of Arts and Sciences (KNAW), the Academia Europea and the (Dutch Government) Advisory Council for Science, Technology and Innovation (AWTI, since 2014). She holds an ERC Advanced grant, a Vici award from NWO and an EBRAINS Lead Scientist Voucher from the Human Brain Project.
Chemistry of the adaptive mind: lessons from dopamine
The human brain faces a variety of computational dilemmas, including the flexibility/stability, speed/accuracy and labor/leisure tradeoff. I will argue that striatal dopamine is particularly well suited to dynamically regulate these computational tradeoffs depending on changing task demands. This working hypothesis is grounded in evidence from recent studies on learning, motivation and cognitive control in human volunteers, using chemical PET, psychopharmacology, fMRI and computational modeling of behavior. These studies also begin to elucidate the mechanisms underlying the huge interindividual variability in the effects of commonly used catecholaminergic drugs. For example, I will demonstrate how effects of the most used psychostimulant methylphenidate on the established monetary incentive delay task can be accounted for by striatal dopamine-dependent changes of specific cognitive parameters derived from a computational drift diffusion model. This modeling allowed us to characterize dopamine’s effects on motivation as reflecting modulation of a race between target-related evidence accumulation and cue-related internal timing processes for affecting the decision about when to respond.
Responsible Research and Innovation in Applied Brain Models
The EMBRACE project seeks to promote Responsible Research and Innovation (RRI) in Future and Emerging Technologies (FET) research through a suite of expert training courses and materials. Applied brain models is an emerging field which raises critical concerns to which responsible research and innovation can be applied. In this session we will discuss two prominent RRI models and draw upon examples of their application in the field of applied brain models.
MULTI-LATERAL: Multi-level Integrative Analysis of Brain Lateralization for Language
Speaker: Prof. dr. Clyde Francks (Max Planck & Donders Institutes & Radboud University Medical Center, Nijmegen)
‘Multi-level Integrative Analysis of Brain Lateralization for Language’ was a HBP partnering project that ran from 2016 to 2019. Some of the main outputs included: 1.The largest genome-wide association studies of different aspects of human structural brain asymmetry (some in over 30,000 individuals). 2. The largest mapping study of brain anatomical correlates of human handedness, linking specific genetic loci to handedness and its brain structural correlates. 3. An atlas of brain regions important for sentence-level language tasks regardless of modality, based on task-based functional activation and resting-state connectivity. 4. Principles of lateralisation and plasticity of the main language systems.
Selected publications:
- Kong et al. Large-scale phenomic and genomic analysis of brain asymmetrical skew. Cerebral Cortex (2021) https://doi.org/10.1093/cercor/bhab075
- Sha et al. The genetic architecture of structural left–right asymmetry of the human brain. Nature Human Behaviour (2021) https://doi.org/10.1038/s41562-021-01069-w
- Sha et al. Handedness and its genetic influences are associated with structural asymmetries of the cerebral cortex in 31,864 individuals, PNAS (2021). https://doi.org/10.1073/pnas.2113095118
- Labache et al. A SENtence Supramodal Areas AtlaS (SENSAAS) based on multiple task-induced activation mapping and graph analysis of intrinsic connectivity in 144 healthy right-handers, Brain Structure & Function (2019) https://doi.org/10.1007/s00429-018-1810-2
- Gurunandan et al. Converging evidence for differential specialisation and plasticity of language systems, J Neurosci (2020) https://doi.org/10.1523/JNEUROSCI.0851-20.2020
- Carrion-Castillo et al. Genetic effects on planum temporale asymmetry and their limited relevance to neurodevelopmental disorders, intelligence or educational attainment, Cortex (2020)
- https://doi.org/10.1016/j.cortex.2019.11.006
Efforts on the integration and large-scale simulation of a whole-brain model led to the development of a collaborative framework for data-driven modeling.
As part of the Japan program for promoting research on the Fugaku supercomputer, we have been working on the integration of biologically grounded models of the cerebral cortex, basal ganglia, cerebellum, and thalamus; as a whole-brain model. We performed resting state simulations for different scales of the integrated network which clarified several issues. Collaboration with NEST developers in the HBP (Human Brain Project) allowed us to improve network construction time and memory consumption, by the incorporation of NEST 3 and new PYNEST functions such as collocated synapses for multiple receptors and aggregated connections rules. Moreover, the collaboration was extended to the NRP (Neurorobotics platform) team in the HBP for integration tests with a biomechanically accurate musculo-skeletal system of rodent, resulting in the development by NRP of a new architecture for large scale co-simulations on the computing resources of EBRAIN.
Models’ building, integration, validation, and large-scale simulations are major challenges. Critical aspects include model transparency and reproducibility, as well as how anatomical and physiological data are efficiently converted into parameters of neurons and synapses, together with the estimation of unknown parameters in reference to experimental observations. In that context, we have developed SNNbuilder, a web-based tool for systematic data-driven development of spiking neural network (SNN) models with focus on remote collaboration. Our framework organizes all data attributes, including species, brain regions, neuron types, projections, neuron models, and references as tables and relations within a database management system (DBMS) and provides GUI interfaces for data registration and visualization. This supports collaborative model building by different users, multiple values for the same parameter and traceability of source information for every detail attached to a model. The framework provides code generation for simulations on NEST 3 and was tested in cortical and striatal network models of various sizes. We have recently deployed SNNbuilder first release on the internet to support the SNN modeling of any region of the brain.

Béchir Jarraya is both neuroscientist and neurosurgeon. He is the leader of the Primate Consciousness and Cognition group at NeuroSpin a research center for neuroscience and neuroimaging south of Paris, France. He is also professor of medicine at Université Paris-Saclay (UVSQ) and head of the Deep Brain Stimulation program at Foch hospital, Suresnes, France. His current interest is to address fundamental questions about brain mechanisms of consciousness and anesthesia-induced loss of consciousness. His group combines non-human primate models, neuroimaging, neuro-stimulation technologies, computational models and deep learning models, to develop, simulate and test new approaches to stimulate the brain and restore consciousness loss.
Deep brain stimulation of the thalamus restores signatures of consciousness in a non-human primate model
Loss of consciousness is associated with the disruption of long-range thalamo-cortical and cortico-cortical brain communication. We tested the hypothesis that deep brain stimulation (DBS) of central thalamus might restore both arousal and awareness following consciousness loss. We modelled unconsciousness using finely tuned anesthesia in primates, a model in which the signatures of loss of consciousness were also found to reflect the clinical status of patients with disorders of consciousness. Under propofol anesthesia, the implanted DBS leads stimulated either central thalamus or a control structure, the ventro-lateral thalamic nucleus. We monitored the effects of DBS on both behavior and brain activity using functional MRI (fMRI) and EEG. We investigated both the cortical dynamics of spontaneous resting-state activity and the brain evoked activity by the “local-global” auditory paradigm. During anesthesia, central thalamic stimulation induced arousal in an on-off manner and increased fMRI activity in prefrontal, parietal and cingulate cortices. Moreover, DBS restored a broad dynamic repertoire of spontaneous resting-state activity, previously described as a signature of consciousness. None of these effects were obtained during the stimulation of the control site in the ventro-lateral thalamus. Finally, DBS restored a broad hierarchical response to auditory violations that was disrupted under anesthesia. Thus, DBS restored the two dimensions of consciousness, arousal and conscious access, following consciousness loss, paving the way to its therapeutical translation in patients with disorders of consciousness.
Comparative Investigation of the Cortical Circuits in Mouse, Non-human primate and Human
CORTICITY required tight collaborations between physiologists, anatomists and theoreticians. Invasive techniques for anatomical and physiological investigations in mouse and macaque allowed the extraction of empirical data and the identification of fundamental principles on cortical organization, both essentials for the development of whole-brain models and large-scale simulations. These whole-brain models simulate various neuronal populations producing different types of functional dynamics in a defined anatomical network. Comparison between simulations from whole-brain models and non-invasive recordings in macaques and humans allowed the identification of functional and structural principles of the cognitive architecture in human and non-human primates subtending different states of consciousness.
Using tract‐tracing and electrophysiological measures of brain activity in mouse and non‐human primate, we have identified architectural principles of the mammalian connectome and how it shapes functional interactions (D’Souza et al. 2022, Nature Communications; Vezoli et al. 2021, Neuron; Froudist-Walsh et al. 2021, Neuron; Hayashi et al. 2021, Neuroimage; Vezoli et al. 2021 Neuroimage; Gămănut et al. 2018, Neuron). Using whole-brain models we have shown that the structure of brain connectivity - specially, the core-periphery architecture and the presence of long-range connections - together with heterogeneous dynamics across brain regions highly determine the brain dynamics at different states of consciousness (Deco et al. 2021, Current Biology; López-González et al. 2021, Nature Communications Biology; Hahn et al. 2021, NeuroImage; Signorelli et al. 2021, NeuroImage). Moreover, the models explain the differences in the brain’s responsiveness and information capacity observed in different brain states of human and animal neural activity (Ponce-Alvarez et al. 2021, Cerebral Cortex; Ponce-Alvarez et al. 2020, eLife; Jobst et al. 2021, NeuroImage). In particular, using tools from statistical mechanics, we showed that transitions between states of consciousness relate to phase transitions and primarily depend on changes in population couplings of specific brain regions (Ponce-Alvarez et al. 2021, Cerebral Cortex). Finally, we identified specific circuit mechanisms for shaping cortex-wide network interactions by key neuromodulatory systems (Pfeffer et al. 2021, Science Advances).
Altogether, these studies contribute to understand how neural network effects sustain different brain states and determine their information capabilities in terms of neural communication, responsiveness, and information capacity.
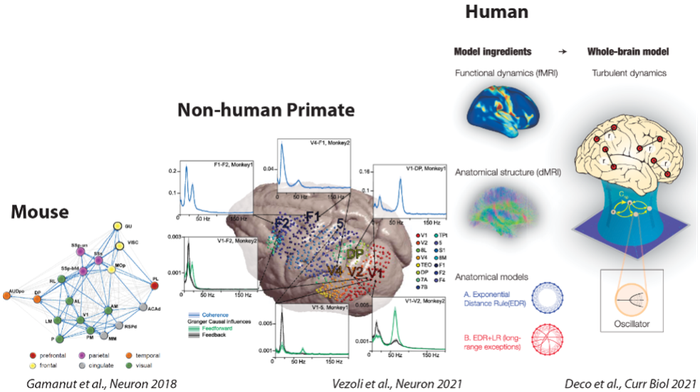
From anatomical and functional connectomes to whole-brain models
How the collaboration between anatomists (H. Kennedy, K. Knoblauch), physiologists (P. Fries, M. Vinck, B. Jarraya) and theoreticians (G. Deco, Z. Toroczkai, M. Ercsey-Ravasz) leads to advances in the understanding of brain function.

Mehdi Khamassi is a research director employed by the Centre National de la Recherche Scientifique (CNRS), and working at the Institute of Intelligent Systems and Robotics (ISIR), on the campus of Sorbonne Université, Paris, France. He has a double background in Computer Science (Engineering diploma in 2003 from Ecole Nationale Supérieure d'Informatique pour l'Industrie et l'Entreprise, Evry with specialization in Artificial Intelligence and Statistical Modeling) and Cognitive Sciences (Cogmaster in 2003 from Université Pierre et Marie Curie (UPMC), Paris). Then he pursued a PhD in Cognitive Neuroscience at UPMC and Collège de France between 2003 and 2007. He serves as co-director of studies for the CogMaster program at Ecole Normale Supérieure (PSL) / EHESS / University of Paris. He also serves as Editor for the several scientific journals, like Intellectica, Frontiers in Neurorobotics, Frontiers in Decision Neuroscience, ReScience X, and Neurons, Behavior, Data analysis and Theory. His main topics of research include decision-making and reinforcement learning in robots and animals, and the role of social and non-social rewards in learning.
Exploiting individual differences to inform computational models of dopamine in reinforcement learning.
The model-free reinforcement learning (MFRL) framework, where agents learn local, cached, implicit action values without trying to estimate a model of their environment, has been successfully applied to Neuroscience in the last two decades. It can account for most dopamine reward prediction error signals (RPEs) in Pavlovian and instrumental tasks. However, it is still not clear why in the Pavlovian autoshaping paradigm RPEs can been recorded in some individuals but not in others. Moreover, the role of dopamine in other functions not related to learning, such as exploration, is still not understood.
Here we present a computational model of dopamine-based learning and exploration regulation, and show how it can account for inter-individual differences, termed sign-tracking and goal-tracking, in a Pavlovian lever-autoshaping procedure. The model combines MFRL with a model-based (MBRL) component which estimates an internal model of different actions’ consequences in the environment. This model can explain inter-individual differences by a different relative contribution weight of MFRL and MBRL in choice behavior: The behavior of sign-trackers is mostly led by MFRL, displaying dopamine RPEs and pushing them towards reward predicting stimuli; In contrast the behavior of goal-trackers is mostly led by MBRL, where dopamine RPEs are absent, and pushing them towards the outcome of their behavioral responses. Moreover, the model suggests that injection of flupenthixol in goal-trackers impairs the exploration-exploitation trade-off and thus blocks the expression of a covert dopamine-independent MBRL learning process.
This model led to a series of novel predictions produced through simulations in variants of the classic autoshaping procedure. Most notably, the model predicts that sign-trackers would be less sensitive to outcome devaluation than goal-trackers, which has recently been confirmed experimentally. The model also predicts that changing the duration of the inter-trial interval should change the relative proportion of sign- and goal-trackers in the population as well as the dopamine RPE profile in these individuals. Recent experimental results collected by the group of Matthew R. Roesch at the University of Maryland, USA, partially confirming these predictions will be presented. Other recent results collected by the group of Etienne Coutureau and Alain Marchand at the CNRS in Bordeaux, also confirm some of the model predictions, namely that dopamine blockage impairs exploration regulation mechanisms in rats.
Together these results suggest a variety of mechanisms on which dopamine can impact, which can be progressively better understood through the tight collaboration between experimentation and computational modeling. Moreover, sign- and goal-tracking behavior having been related to different vulnerability degrees to drug seeking, this computational model may have impacts on the understanding of some individuals’ transition to addiction.
Layer-specific characterization and modeling of fronto-parieto-visual dynamics in primate cortical networks
While visual cortical areas and their interactions have been extensively studied, we know much less about fronto-parietal dynamics in primates. In this project, we aim to reveal how layer- and area-specific network dynamics underlie communication within and between prefrontal, motor, and parietal cortical areas in macaques, and compare these dynamics with those of visual areas. We consider different brain states reflecting different levels of arousal, attention, and predictability during both resting state and behavioral tasks. Spiking data have been collected in three predictive visuo-motor tasks involving hand and/or eye movements. Recordings have been obtained from prefrontal, primary motor, and premotor cortex, parietal areas 7a and DP, and visual areas V1 and V4. The resting-state activity is used to constrain the connectivity of cortical microcircuit models for different areas that are intended as building blocks for a multi-area model encompassing visual, parietal, and frontal regions. To this end, an evolutionary algorithm was customized to fit population-specific connection probabilities to reproduce rates, irregularity, and correlations of spike trains. A data comparison framework was implemented using NetworkUnit and the optimization is demonstrated to work on ground truth data. This forms the basis for constraining layer-resolved microcircuit models of different macaque cortical areas, to be incorporated into a multi-area model that enables studying visuo-motor interactions.
Map of dopamine 1- and 2-receptor positive cells in the developing mouse forebrain:perceived fit of registered atlas across sex, age, and regions
Bjerke IE1,Charan KM2, Leergaard TB1, Kim JH3*
Affiliations:
- Neural Systems Laboratory, Institute of Basic Medical Sciences, University of Oslo, Oslo, Norway
- ISN Psychology, Institute for Social Neuroscience, Ivanhoe, Australia;
- IMPACT – the Institute for Mental and Physical Health and Clinical Translation, School of Medicine, Deakin University, Geelong, VIC, Australia
The dopaminergic system undergoes major changes throughout development, which may contribute to vulnerability to mental disorders early in life. Forebrain neurons expressing dopamine 1 and 2 receptors (D1R and D2R, respectively) are particularly sensitive to dopaminergic input. Understanding their typical development is helpful in studying the ontogeny of different mental disorders. With support from the Human Brain Project voucher DOPAMAP, we have digitized and organized a comprehensive collection of microscopic images of immunostained D1R and D2R expressing neurons across the forebrain from 152 male and female mice at five stages of life (P17, P25, P35, P49, and adult). All images are registered to the Allen Mouse brain Common Coordinate Framework. For the P17-P35 age groups, the atlases were spatially modified to match the morphology of young brains. Rodent Brain Atlases are critical tools to understand normative brain structure. However, no female rodent atlases exist despite the presence of sexual dimorphism. Therefore, a scorer blind to group allocations judged the perceived size and position fit of the P17-P35 brain images against assigned atlases, focusing on the nucleus accumbens, olfactory tubercle, and the caudate putamen. We hypothesized that the registration of female mouse brains to a male mouse brain atlas would result in lower perceived fit as the age of the brains increased due to emerging sexual dimorphism. Overall, perceived fit was significantly lower when the brains were from younger mice. Sex difference was only observed for anterior nucleus accumbens position, with females showing a poorer fit than males. These results assure that sex differences in mouse atlas fit are likely marginal. In contrast, using atlas for developing mice calls for caution, and any attempts to target specific brain regions in developing mice likely require initial neuroanatomical pilot study. The high-resolution images are shared via an interactive web-microscopy viewer and as downloadable files via the EBRAINS Knowledge Graph. Spanning five age groups and including both male and female subjects, DOPAMAP collection is an important resource for any researcher interested in studying forebrain regions with D1R and D2R positive cells.
 Petra Ritter heads the Brain Simulation Section at the Dept. of Neurology, Charité and Berlin Institute of Health. She is a full professor for Brain Simulation. Her research focus is on integrating neuroimaging and computational neuroscience to discover mechanisms of brain function and dysfunction. She has led local (Charité & BIH Virtual Research Environment[1]), national (Bernstein Focus), European (VirtualBrainCloud; Co-design Project The Virtual Brain in the EU Flagship HBP SGA2) and international neuroiformatics consortia (co-lead The Virtual Brain platform with >30k software downloads), is a participant in the national research data infrastructure initiative (NFDI) NFDI4Health and a co-speaker in NFDI-Neuro and a member of the European Open Science Cloud Architecture working group. She holds an ERC Consolidator grant and is engaged in several large-scale software projects. In the €15Mill funded EU project Virtual Brain Cloud and its local partnering project Virtual Research Environment, Ritter and collaborators develop a reference architecture for cross-institutional managing and processing of radiologic imaging and associated data in compliance with GDPR. Ritter’s Brain Simulation Section is GOLD member of the International Neuroinformatics Coordination Facility (INCF). Petra Ritter studied medicine at the Charité University Medicine Berlin. She spent a large part of her clinical traineeships and practical year abroad: at the universities UCLA and UCSD in Los Angeles and San Diego, the Mount Sinai School of Medicine in New York and the Harvard Medical School in Boston. In 2002, she received her license to practise medicine. In 2004, she completed her doctoral thesis at the Charité and in 2010 she received habilitation in Experimental Neurology. After being Max Planck Minerva research group leader from 2011 to 2015, she assumed the lifetime position of BIH Johanna Quandt Professor for Brain Simulation at Berlin Institute of Health (BIH) and Charité Universitätsmedizin Berlin, one of Europe’s largest university hospitals. Since 2017, she is Director of the Brain Simulation Section at Charité Universitätsmedizin Berlin.
Petra Ritter heads the Brain Simulation Section at the Dept. of Neurology, Charité and Berlin Institute of Health. She is a full professor for Brain Simulation. Her research focus is on integrating neuroimaging and computational neuroscience to discover mechanisms of brain function and dysfunction. She has led local (Charité & BIH Virtual Research Environment[1]), national (Bernstein Focus), European (VirtualBrainCloud; Co-design Project The Virtual Brain in the EU Flagship HBP SGA2) and international neuroiformatics consortia (co-lead The Virtual Brain platform with >30k software downloads), is a participant in the national research data infrastructure initiative (NFDI) NFDI4Health and a co-speaker in NFDI-Neuro and a member of the European Open Science Cloud Architecture working group. She holds an ERC Consolidator grant and is engaged in several large-scale software projects. In the €15Mill funded EU project Virtual Brain Cloud and its local partnering project Virtual Research Environment, Ritter and collaborators develop a reference architecture for cross-institutional managing and processing of radiologic imaging and associated data in compliance with GDPR. Ritter’s Brain Simulation Section is GOLD member of the International Neuroinformatics Coordination Facility (INCF). Petra Ritter studied medicine at the Charité University Medicine Berlin. She spent a large part of her clinical traineeships and practical year abroad: at the universities UCLA and UCSD in Los Angeles and San Diego, the Mount Sinai School of Medicine in New York and the Harvard Medical School in Boston. In 2002, she received her license to practise medicine. In 2004, she completed her doctoral thesis at the Charité and in 2010 she received habilitation in Experimental Neurology. After being Max Planck Minerva research group leader from 2011 to 2015, she assumed the lifetime position of BIH Johanna Quandt Professor for Brain Simulation at Berlin Institute of Health (BIH) and Charité Universitätsmedizin Berlin, one of Europe’s largest university hospitals. Since 2017, she is Director of the Brain Simulation Section at Charité Universitätsmedizin Berlin.
Virtual Brain Cloud: Enabling Complex Simulations with Multilevel Health Data
Virtual Brain Cloud generates multilevel virtual brains of patients and healthy human controls for research and innovation. Brain data from multiple sourcesare being pre-processed and annotated with a common data model - such that they all relate to common spatial reference frameworks. The platform thus offers a next generation clinical research infrastructure - compliant with Data Protection Regulations and creates an open yet protected space for groundbreaking digital health innovation.
Integrated effects of multiple Attentional Control signals in the primate brain
or
MAC-Brain
Developing a Multi-scale account of Attentional Control as the constraining interface between vision and action: A cross-species investigation of relevant neural circuits in the human and macaque Brain
Santandrea E1, Ben-Hamed S2, Boehler NC3, Chelazzi L1, Macaluso E4
- University of Verona, Verona, Italy
- Institut des Sciences Cognitives Marc-Jeannerod, Lyon, France
- Ghent University, Ghent, Belgium
- Lyon Neuroscience Research Center, Lyon, France
Mechanisms of Attentional Control (AC) in primates have been extensively investigated, especially in humans, showing that several factors determine how the brain selectively processes incoming sensory information and targets motor output to behaviorally relevant objects in the environment. AC can be considered as an overarching mechanism that integrates signals from multiple cognitive domains in order to assign processing priorities to specific sensory inputs and gate motor response selection in the service of goal-oriented behavior. Available data already identified neurophysiological correlates of different factors contributing to AC, but the highly specialized nature of most experiments has prevented a systematic study of the interactions between these factors; thus, it remains unclear whether different AC signals operate synergistically or independently in assigning attentional priority to a specific item/location.
The MAC-Brain Partnering Project was aimed at addressing this issue directly and systematically via an integrated and multidisciplinary approach, developed to characterize the unique and combined influence of different AC mechanisms, i.e. goal-directed AC, bottom-up mechanisms and experience-dependent biasing signals derived from a statistical learning process. Specifically, we developed variants of a visual search task where participants had to find the target and report the position of a small gap (up/down) within it. Within series of experiments using the same task design with different techniques (behavior, EEG, fMRI, electrophysiology), we manipulated multiple AC signals at a time and directly studied the resulting interactions. Overall, a predominance of the top-down biasing signal emerged over both bottom-up and experience-dependent signals: while, considering behavior, this prevalence might even take the form of a full gating, preventing other AC signals to manifest their influence, at the neural level the latter still have an influence on ongoing processing of information. Interestingly, we also unveiled that the level at which different AC signals interact in the brain might vary considerably: for example, the joint influence of perceptual salience and top-down guidance seems to be fully specified within the occipital visual cortex, while the mechanisms controlling spatial selection based on statistical regularity possibly rely on higher-order spatial representations rather than the modulation of early processing. Albeit to be finalized, collected evidence represents a first significant stride towards a deeper understanding of the functional and neural integration of different AC signals in the primate brain.
Integrative Frameworks in Single-cell Connectomics
The past decade has seen a tremendous development of connectomics approaches, with single-neuron reconstructions bridging the gap between cellularly resolved micro-circuits and whole-brain bulk axonal projections. However, characterizing brain-wide projection motifs in thousands of neurons is still challenging. We have thus developed a tool for translating ~3000 axonal morphologies into projection statistics across different brain areas, using reconstructed neurons from high throughput repositories and from our recently developed pipeline.
We first identify neurons with similar axonal morphology and registration coordinates using an in-house neuron as template. Particularly, we apply the Coherent Point Drift method to compute similarity by first finding correspondences between points in target and template neuron followed by determining the mean squared distance between corresponding points. Given the group of most similar neurons, we use the Allen Reference Atlas to measure the distribution of axonal length across cortical layers and subcortical nuclei We obtained projection statistics for a group of similar thalamic neurons showing high axonal overlap in layers 2-5 of the lateral visual area and in various thalamic nuclei. In addition, we repeated the procedure with the more densely sampled intra-cortical projection neurons, however results are still preliminary.
Our approach represents a significant extension to existing tools, since it accounts for full axonal trees instead of only regions of interest. Moreover, it is applicable to neurons reconstructed in smaller laboratories and allows for integration with other similarly registered datasets. We further intend to extend our approach to uncover complex projection motifs. Lastly, we are currently working on simulating the action potential propagation across the axons of the above thalamocortical neurons using multi-compartmental models from the NEURON simulation environment. The primary motivation of the simulations is to understand the relationship between the complexity of long-range axonal trees and delays in the propagation of the action potential.
Project FIIND: Ferret Interactive Integrated Neuro Development Atlas
The first days after birth in ferrets provide a unique view of the development of a complex brain. Unlike mice, ferrets develop a rich pattern of deep neocortical folds and cortico-cortical connections. Unlike humans and other primates, whose brains are well differentiated and folded at birth, ferrets are born with a very immature and completely smooth neocortex: folds, neocortical regionalisation and cortico-cortical connectivity develop in ferrets during the first days after birth. Project FIIND aimed at creating the most detailed description of the development of the ferret brain. We acquired data during the first days of ferret brain development using multiple MRI modalities and whole brain histology. Through FIIND's website researchers can acces our data, visualise it, and work with it directly in the Web browser. In addition to the interactive user interface, raw data and annotations can be accessed programmatically, and we provide Jupyter notebooks demonstrating the procedure.
CerebNEST: A Bioinspired Multiscale Modelling of The Cerebellar Network
CerebNEST project aims at building a large-scale neural network model of the cerebellum as point-neuron detailed circuit (in NEST).
The network comprises cell-specific neuron models and plastic connections, and each cell is placed and connected using the Brain Scaffold Builder according to a set of physiological rules extracted according to morphology and anatomical information. This cerebellar network model can be tested as a stand-alone circuit, or it can be embedded in a virtual robot and perform closed-loop simulations. Towards this last aim, additional functional neuron models have been developed to complete the closed-loop controller. Specifically, we defined and implemented an analog-to-spiking neuronal interface to encode kinematic and dynamic signals and a state estimation neuron model performing a bayesian integration between the sensory feedback and the cerebellar prediction. The embedding and simulations of the full cerebellar model in neurorobotic platforms result in a considerable computational load. For this reason, we are currently exploiting HPC resources to meet this criterion and working towards simplified pipelines reducing the computational load without losing model realism.
We are also implementing a new feature that will allow us to simulate the role of the Nitric Oxide as a diffusive second messenger regulating plasticity mechanisms.
Layer-specific characterization and modeling of fronto-parieto-visual dynamics in primate cortical networks
While visual cortical areas and their interactions have been extensively studied, we know much less about fronto-parietal dynamics in primates. In this project, we aim to reveal how layer- and area-specific network dynamics underlie communication within and between prefrontal, motor, and parietal cortical areas in macaques, and compare these dynamics with those of visual areas. We consider different brain states reflecting different levels of arousal, attention, and predictability during both resting state and behavioral tasks. Spiking data have been collected in three predictive visuo-motor tasks involving hand and/or eye movements. Recordings have been obtained from prefrontal, primary motor, and premotor cortex, parietal areas 7a and DP, and visual areas V1 and V4. The resting-state activity is used to constrain the connectivity of cortical microcircuit models for different areas that are intended as building blocks for a multi-area model encompassing visual, parietal, and frontal regions. To this end, an evolutionary algorithm was customized to fit population-specific connection probabilities to reproduce rates, irregularity, and correlations of spike trains. A data comparison framework was implemented using NetworkUnit and the optimization is demonstrated to work on ground truth data. This forms the basis for constraining layer-resolved microcircuit models of different macaque cortical areas, to be incorporated into a multi-area model that enables studying visuo-motor interactions.
SENSEI: uncovering neuron structure using hard and soft approaches
SENSEI aims at developing new tools for neuron segmentation at different levels, ranging from neuronal shape to finer structures such as dendritic spines. In this presentation, we will introduce tissue processing and labeling strategies to deal with thick ex-vivo brain slices, as well as a new segmentation tool exploiting both topological and image intensity information. The first results about a new fast super resolution-compatible imaging system based on line-scan confocal imaging device will be presented.
Computational analysis of primate vision
Primates, especially macaque monkeys, are well established model organisms for human vision. The main goal of our research is to combine the biological and the computational approaches and create the first physiologically plausible working model of early cortical vision in primates.
Our main tool, CxSystem2 (Andalibi et al., 2019), was introduced to Human Brain Project 2019. This tool allows efficient simulations of small and medium-sized cortical models.
Earlier, we have published an extensive review on macaque visual cortical areas V1—the primary visual cortex—and areas V2 and V5 (Vanni et al., 2020). We have ongoing work on primate retina model, based on data from Chichilnisky lab (Field et al., 2010). Recently, we have followed the work by Brendel et al.(2020) who showed how a spiking network can efficiently and robustly learn to represent a continuous input signal. We complement their work a step closer to biology and quantify how basic membrane properties affect information transfer.
Future open questions include abstraction of key pyramidal cell apical dendrite nonlinearities as well as implementation of plasticity mechanisms between distinct cell types. For a layered system, we plan to leverage a working model from Grossberg et al. (2008). Area summation function is likely to become our first computational target, given its role in combining contextual to receptive field center data as well as abundant existing data on its mechanisms (Angelucci et al., 2017).
References:
Andalibi V, Hokkanen H, Vanni S (2019) Controlling Complexity of Cerebral Cortex Simulations-I: CxSystem, a Flexible Cortical Simulation Framework. Neural Comput 31:1048–1065.
Angelucci A, Bijanzadeh M, Nurminen L, Federer F, Merlin S, Bressloff PC (2017) Circuits and Mechanisms for Surround Modulation in Visual Cortex. Annu Rev Neurosci 40:425–451.
Brendel W, Bourdoukan R, Vertechi P, Machens CK, Denève S (2020) Learning to represent signals spike by spike. PLoS Comput Biol 16.
Field GD, Gauthier JL, Sher A, Greschner M, MacHado TA, Jepson LH, Shlens J, Gunning DE, Mathieson K, Dabrowski W, Paninski L, Litke AM, Chichilnisky EJ (2010) Functional connectivity in the retina at the resolution of photoreceptors. Nature 467:673–678.
Gerstner W, Kistler WM, Naud R, Paninski L (2014) Neuronal dynamics: From single neurons to networks and models of cognition. Cambridge: Cambridge University Press.
Grossberg S, Versace M (2008) Spikes, synchrony, and attentive learning by laminar thalamocortical circuits. Brain Res 1218:278–312.
Vanni S, Hokkanen H, Werner F, Angelucci A (2020) Anatomy and physiology of macaque visual cortical areas V1, V2 and V5/MT: bases for biologically realistic models. Cereb Cortex 30:3483–3517.
Organising Committee
Scientific Chair & Local Host
Julien Vezoli | Ernst Strüngmann Institute (ESI) for Neuroscience in Cooperation with Max Planck Society
Paul Verschure | Donders Institute for Brain, Cognition and Behaviour
PP Coordinators
Marie-Elisabeth Colin | EBRAINS, HBP Partnering Task Leader
Angeliki Lorents | EBRAINS
Supported by

In cooperation with



Venue
Fletcher Landgoed Hotel Holthurnsche Hof
Zevenheuvelenweg 48A
6571 CK Berg en Dal, Netherlands





















